Hello, learning team The requirements of the FDA at the moment (31/10/2017) are as follows: Ingredients, groups that are not controlled substances, use rate can be notified without telling the rate of use. (however If a member wants to disclose to the FDA, they can reveal it. It's not wrong, but most manufacturers or brand owners will choose not to disclose. To conceal trade secret information.) Ingredients. The rate of use is controlled (FDA is set to use no more than a few % in each item, such as
สารกันเสีย Various Colors
สารกันแดด various) must disclose the rate of use in the formula. in the notice document For the ingredients that members inquire about There will be items 1,2 that need to be computed separately. And disclose the usage rate of each component with the FDA. Members can request MSDS documents from the customer relationship
care@myskinrecipes.com which way Customer relations will send documents. There will be a topic that describes the composition of that substance. Similar to the picture below. which is a component of
Phenoxyethanol SA [attachment=0: 2e371n35]
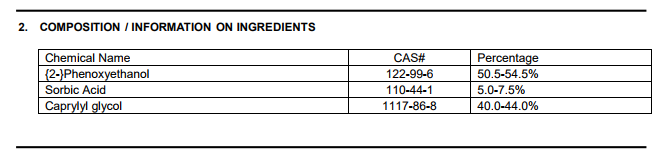
- 2017-10-31_1-08-29.png (13.72 KiB) Viewed 1627 times

