Cosmetic Formulation Compatibility and Stability Issues
Asked by: vichai_chan
On: September 10, 2022
Product Type:
Cosmetics
Answered
Question
I have a cosmetic formulation with various active ingredients. Could you please review the ingredient list for any potential compatibility or stability issues?
Answer
Compatibility Review of Your Formulation
Your formulation contains a variety of beneficial active ingredients aimed at antioxidant protection, skin brightening, and barrier support. However, there are significant compatibility challenges that need to be addressed for the formula to be stable and effective.
Key Compatibility Issues:
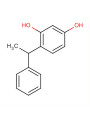
- Severe pH Mismatch: This is the most critical issue. The formula contains ingredients that require an acidic or slightly acidic pH for stability and efficacy, such as Ethyl Ascorbic Acid (Vitamin C Ethyl Ether) (optimal pH 3.5-6.0) and Phenylethyl Resorcinol (optimal pH 4-5). At the same time, the formula includes 1% Sodium Hydroxide, which is a strong base. Adding 1% Sodium Hydroxide will raise the pH significantly, likely far above the optimal range for these acidic actives. A high pH will cause rapid degradation of Ethyl Ascorbic Acid and Phenylethyl Resorcinol, rendering them ineffective and potentially causing discoloration.
- CysWhite™ (Cysteamine HCL) in a Leave-on Formula: Cysteamine HCL (CysWhite™) at a concentration of 5% is typically used as a short-contact (wash-off) treatment for hyperpigmentation due to its strong odor, potential for irritation, and instability in water-based formulas, especially at certain pH levels. While ActiveProtec™ OX is included to help with oxidation, maintaining the stability and managing the strong odor of 5% Cysteamine HCL in a water-based leave-on product with other reactive ingredients and pH issues is highly challenging. The product description for CysWhite™ explicitly recommends washing it off after 15-30 minutes and suggests anhydrous formulas for long-term stability.
- Emulsion and Gel Stability: The formula is a complex mix of water-soluble and oil-soluble ingredients, along with alcohol and potential electrolytes from ingredients like Dipotassium Glycyrrhizate, Sodium Hydroxide, ActiveProtec™ OX, and CysWhite™ HCL. While LecithinGel™ and Phospholipid are included as binders/emulsifiers, the presence of alcohol and electrolytes, combined with extreme pH fluctuations caused by Sodium Hydroxide, can easily destabilize the emulsion and the gel structure provided by Xanthan Gum.
- Ingredient Interactions: Phenylethyl Resorcinol can react with amino acids, urea, and certain proteins, potentially causing discoloration or instability. While these are not explicitly listed, some complex ingredients like Skin-Barrier™ or RoseShield™ could contain components that interact. Ethyl Ascorbic Acid should ideally avoid Vitamin B3 and Zinc PCA. The high concentration of multiple antioxidants (Vitamin E, Astaxanthin, ROE, EUK-134, RoseShield™, Pure-EGCG™, ActiveProtec™ OX) could also lead to complex interactions, although they are generally beneficial.
Recommendations:
- Address the pH Conflict: The most critical step is to resolve the pH incompatibility. It is highly unlikely that all the active ingredients in this formula can be stable and effective at a single pH. You may need to:
- Reformulate into separate products for incompatible actives (e.g., one formula for acidic actives, another for actives stable at higher pH).
- Significantly reduce or eliminate Sodium Hydroxide and carefully adjust the pH to a range that favors the most critical actives, accepting that others may be less effective or unstable. However, achieving stability for both acidic actives and CysWhite™ in the same water-based formula is very difficult.
- Reconsider CysWhite™ Usage: Using 5% CysWhite™ in a leave-on formula is not recommended due to stability, odor, and potential irritation. Consider using a lower concentration, formulating it in an anhydrous base, or using it in a wash-off product as suggested by the manufacturer.
- Stability Testing: Due to the complexity and potential incompatibilities, rigorous stability testing (including pH monitoring, viscosity, appearance, and active ingredient concentration over time and under different temperature conditions) is absolutely essential.
- Review Trademarked Ingredient Details: Obtain detailed information on the composition and compatibility guidelines for trademarked ingredients like Mild Preserved Eco™, Skin-Barrier™, RoseShield™, LecithinGel™, and ActiveProtec™ OX to identify any specific contraindications. (Note: Specific details for RoseShield™ were not available in our database).
- Check Vitamin E Duplication: Confirm if Vitamin E is intended to be at 1% or 2% total, as it is listed twice at 1%.
In summary, while the formula contains many promising ingredients, the current composition, particularly the pH conflict and the inclusion of 5% CysWhite™ in a likely leave-on, water-based system, presents significant stability and efficacy challenges.