DMI Compatibility with Encapsulated 4-Butylresorcinol in Whitening Cream
Question
I am developing a whitening cream for freckles with the following key ingredients:
- DMI (Dimethyl Isosorbide) 3%
- Encapsulated 4-Butylresorcinol
- Tranexamic Acid
- Niacinamide
- Allantoin
- Ceramide
- Matrixyl 3000
- Retinal-EZ
My primary concern is: Will using DMI at 3% concentration potentially disrupt or destroy the encapsulation structure of the Encapsulated 4-Butylresorcinol (e.g., liposomal system)?
Answer
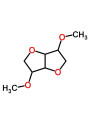
Compatibility of DMI (3%) with Encapsulated 4-Butylresorcinol
The use of Dimethyl Isosorbide (DMI) at a concentration of 3% in a formulation containing Encapsulated 4-Butylresorcinol (such as NanoRes™, which is a liposomal system) poses a potential risk of compromising the encapsulation structure.
- DMI's Role: DMI is a powerful, non-ionic solvent and penetration enhancer. Its primary function is to increase the solubility and skin penetration of active ingredients.
- Encapsulation Structure: Encapsulated actives, especially those using liposomes (lipid-based vesicles), rely on a delicate lipid bilayer structure to protect the active and provide controlled release.
- The Risk: DMI's strong solvent properties can potentially interact with and disrupt the lipid components of the liposome wall. This disruption can lead to:
- Premature Release (Leaking): The 4-Butylresorcinol may "leak" out of the liposome before application, negating the benefits of encapsulation (stability, controlled release, reduced irritation).
- Loss of Stability: The released active ingredient may become less stable in the final cream base compared to when it was protected inside the capsule.
Recommendation:
- Consult the Supplier: The most reliable source of information is the supplier of the specific Encapsulated 4-Butylresorcinol (e.g., NanoRes™). They often provide compatibility data with common cosmetic solvents like DMI.
- Test Compatibility: If compatibility data is unavailable, a small-scale stability test is mandatory.
- Method: Mix the DMI (3%) with the encapsulated active in a small sample of the final base/solvent system.
- Observation: Observe the mixture immediately and over a period of 24-48 hours for any signs of phase separation, cloudiness, or change in viscosity, which could indicate a structural breakdown.
- Long-Term Stability: For a commercial product, accelerated stability testing (e.g., at elevated temperatures) is required to ensure the encapsulation remains intact throughout the product's shelf life.
- Alternative Solvents: If DMI proves incompatible, consider using gentler solvents or humectants like Propanediol or Butylene Glycol, which are less likely to disrupt liposomal structures.
Other Formulation Considerations:
The overall formula is highly potent for treating melasma/freckles. Ensure the final pH is appropriate for all actives:
- Tranexamic Acid & Niacinamide: Generally stable across a wide range, but Niacinamide is best around pH 5.0-7.0.
- Retinal-EZ (Retinaldehyde): Requires a slightly acidic to neutral pH (typically 5.5-6.5) for optimal stability.
- 4-Butylresorcinol: Stable in a wide pH range.
The combination of Retinal-EZ and 4-Butylresorcinol is powerful and may increase the risk of irritation. The inclusion of Allantoin and Ceramide is excellent for mitigating this irritation and supporting the skin barrier. Matrixyl 3000 is a peptide for anti-aging and should be compatible with the other ingredients.