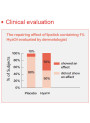
Emulsifying Vitamin E and Formulation Challenges in Serum
Question
I am formulating a serum containing Vitamin E, Ferulic Acid, L-Ascorbic Acid, and Zinc PCA. I need advice on how to properly emulsify Vitamin E into the water phase. Should I use Polysorbate 20 or Polysorbate 80? What is the recommended ratio of emulsifier to Vitamin E? Additionally, I am concerned about the solubility of Ferulic Acid and the compatibility of L-Ascorbic Acid and Zinc PCA regarding pH requirements in the same formula. Could you provide guidance on these formulation challenges?
Answer
Choosing an Emulsifier for Vitamin E
When emulsifying an oil like Vitamin E into a water-based formula, you need an oil-in-water (O/W) emulsifier. The effectiveness of an emulsifier for a specific oil depends on its Hydrophilic-Lipophilic Balance (HLB) value. O/W emulsifiers typically have high HLB values.
- Polysorbate 20 (Tween 20) has an approximate HLB value of 16.7. It is soluble in water and glycerin.
- Polysorbate 80 (Tween 80) has an approximate HLB value of 15.0. It is soluble in both oil and water but not in glycerin.
For emulsifying oils like Vitamin E into a water phase, emulsifiers with higher HLB values are generally more effective. Therefore, Polysorbate 20 is typically a more suitable choice than Polysorbate 80 for emulsifying Vitamin E in an O/W system due to its higher HLB.
The suggestion from the previous reply to use a 2:1 ratio of emulsifier to oil (e.g., 6g of Tween 20 for 3g of Vitamin E) is a common starting point for solubilizing or emulsifying oils with polysorbates. You may need to adjust this ratio based on the specific type of Vitamin E you are using and to achieve the desired clarity and stability of your final product.
Considerations for Your Formula
Based on the previous conversation and the ingredients in your proposed formula, here are a few additional points:
- Ferulic Acid Solubility: As mentioned previously, Ferulic Acid is not soluble in water. It requires a solvent like Ethoxydiglycol to dissolve properly in your formulation. Without a suitable solvent, the Ferulic Acid will not be fully incorporated and may precipitate.
- pH Compatibility: Your formula includes L-Ascorbic Acid, which requires a low pH (ideally 3.5-4.0) for stability and effectiveness. Zinc PCA, however, is most soluble and stable in a pH range of 4-6. Adjusting the pH to 4.5 might be a compromise, but it's important to monitor the stability and clarity of the solution over time, as the lower pH required for Vitamin C could potentially affect the solubility of Zinc PCA.
By addressing the solubility of Ferulic Acid and considering the pH requirements of your active ingredients, you can improve the stability and efficacy of your serum formulation.