Eye Cream Formulation Issues: Pilling, Viscosity Loss, and Carbomer Neutralization
Question
Regarding the eye cream formulation, I have the following questions:
- What causes the issue where the cream rolls off or forms clumps like "ขี้ไคล" (dead skin/residue) when applied?
- Is the method of preparing
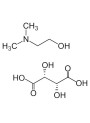
Pro Polymer Silk (Polyquaternium-37)by stirring, refrigerating for hydration, and then stirring again, a correct way to handle this polymer? - Why does the gel lose viscosity and thin down after adding ingredients such as
Gotukola ext.,Repair Activator,NAG, andMSH-white? - Is it true that adding solutions with high electrolyte concentrations, like
NaOH 1MorNaHCO3, to adjust pH can cause gels or creams thickened with electrolyte-sensitive systems (likePro Polymer Silk) to thin down? - Could you explain why
TEAandNaOHare commonly recommended bases for neutralizingCarbomerto build viscosity, whyKOHcan work but is less common, and whyNa2CO3is generally not suitable for this purpose?
Answer
Addressing Your Eye Cream Formulation Issues
Thank you for reaching out and providing details about your eye cream formulation and the problems you've encountered. It's great that you're experimenting and seeking to understand the science behind it. Let's break down your questions:
1. The "Pilling" or Clumping Issue
Cause: The issue where the cream rolls off or forms clumps like "ขี้ไคล" (dead skin/residue) is most likely due to an incompatibility between the cationic polymer, Pro Polymer Silk (Polyquaternium-37), and anionic ingredients. Cationic polymers have positive charges, while anionic ingredients have negative charges. When mixed or applied together, they can form insoluble complexes that precipitate and cause pilling on the skin.
Looking at your formula, there are no obvious anionic ingredients listed. However, this pilling often occurs when the product is applied over other cosmetic products (like serums, toners, or moisturizers) that contain common anionic emulsifiers or gelling agents. The interaction happens on the skin surface during application.
How to fix:
- Test on clean skin: Try applying your eye cream on completely clean, dry skin without any other products underneath. If the pilling stops, the issue is definitely due to incompatibility with another product you are using.
- Layering: If external interaction is the cause, try waiting longer between applying the previous product and your eye cream, or try applying the eye cream first.
- Reformulation (less likely): If pilling still occurs on clean skin, there might be a subtle incompatibility within your formula or the concentration of Pro Polymer Silk is too high. Reducing the Pro Polymer Silk slightly might help, but external product interaction is the more common cause for this type of pilling with cationic polymers.
2. Preparation of Pro Polymer Silk
Your method of preparing the Pro Polymer Silk solution by stirring, refrigerating for hydration, and then stirring again is a standard and correct way to handle this type of polymer. It allows the polymer to fully hydrate and develop its viscosity. Using a magnetic stirrer is also appropriate for laboratory-scale mixing.
3. Loss of Viscosity After Adding Ingredients
This is expected behavior for Pro Polymer Silk (Polyquaternium-37). As a cationic polymer, its viscosity is very sensitive to electrolytes (salts). When you add ingredients that contain ions (like salts of Sodium, Potassium, Magnesium, Calcium, etc.), these ions interfere with the positive charges on the polymer chains, reducing the electrostatic repulsion that causes thickening. This leads to a decrease in viscosity.
Looking at your ingredients, several could contribute electrolytes:
- Gotukola ext. (Centella Asiatica Extract): Plant extracts often contain mineral salts.
- Repair Activator (Bifida Ferment Lysate): Ferment lysates are complex mixtures that can contain various ions and metabolites.
- Potentially NAG (N-Acetyl Glucosamine) or MSH-white (Undecylenoyl Phenylalanine) depending on their specific form or purity.
These electrolytes are the likely reason your gel thinned down after adding these ingredients.
4. Electrolytes and Viscosity Reduction (Pro Polymer & Emulsifiers)
You are absolutely correct. Electrolytes interfere with the thickening mechanism of many gelling agents and emulsifiers, especially those that rely on ionic charges (like anionic Carbomer or cationic Polyquaternium-37, or many ionic emulsifiers). Adding a solution with a high concentration of ions, like NaHCO3 or NaOH 1M, to adjust pH will indeed likely cause a gel or cream thickened with an electrolyte-sensitive system to thin down. The ions shield the charges on the polymer/emulsifier, reducing the repulsive forces needed for the structure (gel or emulsion stability) to hold.
5. Carbomer Neutralization Bases (TEA, NaOH vs. KOH, Na2CO3)
Scientific Explanation: Carbomer is an acidic polymer that thickens when its acidic groups (-COOH) are neutralized by a base. This neutralization creates negatively charged carboxylate groups (-COO⁻) along the polymer chain. These negative charges repel each other, causing the polymer to uncoil and expand, forming a gel structure that traps water.
Why TEA and NaOH are commonly recommended:
- NaOH (Sodium Hydroxide): A strong base that effectively and completely neutralizes the Carbomer's acidic groups, generating maximum negative charges and achieving high viscosity. It's a standard, effective, and cost-efficient choice.
- TEA (Triethanolamine): An organic amine base. It also effectively neutralizes Carbomer and is popular in cosmetics because it provides a more gradual and easier-to-control pH adjustment compared to strong inorganic bases. It typically yields clear gels.
Why KOH (Potassium Hydroxide) can work but is less common: KOH is also a strong base, very similar to NaOH. Scientifically, it can be used to neutralize Carbomer effectively. The reason it might be less commonly recommended than NaOH could be due to slight differences in resulting gel properties (clarity, feel, stability), cost, regional availability, or simply historical preference in cosmetic formulation guides. It is a viable option, but NaOH and TEA are more standard.
Why Na2CO3 (Sodium Carbonate) is less suitable: Na2CO3 is a weak base. It cannot fully neutralize all the acidic groups on the Carbomer polymer. This results in fewer negative charges, less repulsion, and significantly lower viscosity compared to using a strong base like NaOH or TEA. Additionally, the reaction can release CO2 gas, causing foaming, and the carbonate ion might interfere with the gel structure.
In summary: For optimal Carbomer thickening, you need a base strong enough to fully neutralize the acidic groups (like NaOH, KOH, or TEA). NaOH and TEA are the most standard choices in cosmetics for practical formulation reasons, although KOH is scientifically capable. Weak bases like Na2CO3 are generally not effective for achieving significant viscosity with Carbomer.