Jelly Soap Gelling Issue with Kappa Carrageenan
Question
I am trying to make a bouncy and firm jelly soap using Kappa Carrageenan at 1.5% or 2% concentration in a formula containing DI Water, 30% Surfactant, 15% Glycerin, 0.2% Disodium EDTA, and 1% Phenoxyethanol. However, the mixture remains liquid and does not gel. Is Kappa Carrageenan suitable for jelly soap, what concentration should I use, and can you advise on the formula to achieve the desired bouncy and firm texture?
Answer
Jelly Soap Formulation with Kappa Carrageenan
Thank you for your question regarding your jelly soap formulation using Kappa Carrageenan. It sounds like you're aiming for a bouncy, firm texture, but the formula isn't gelling as expected at 1.5% and 2% Kappa Carrageenan.
Kappa Carrageenan is indeed a suitable gelling agent and can create firm gels, which is why it's used in jelly soap. However, Kappa Carrageenan requires the presence of specific ions, particularly potassium (K⁺) or calcium (Ca²⁺) ions, to properly hydrate and form a strong gel structure. Looking at your formula, it appears you don't have an ingredient that provides these necessary ions.
Your formula includes:
- DI Water
- Kappa Carrageenan (1.5% / 2%)
- Surfactant (30%)
- Glycerin (15%)
- Disodium EDTA (0.2%)
- Phenoxyethanol (1%)
The high concentration of surfactant (30%) and Glycerin (15%) can also interfere with the gelling process, making it harder for the Kappa Carrageenan to hydrate and form a stable network, even if ions are present.
To help your jelly soap gel, you need to add a source of potassium or calcium ions to the formula. Common options include:
- Sodium Chloride (Table Salt): While primarily a source of sodium ions, it can help, though potassium is generally more effective for Kappa Carrageenan.
- Potassium Chloride
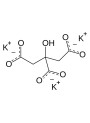
- Potassium Citrate
Try adding a small amount (e.g., 0.5% to 1%) of an ionic compound like Sodium Chloride or Potassium Citrate to your water phase before adding the Kappa Carrageenan. This should provide the necessary ions for gelling.
Regarding the concentration of Kappa Carrageenan, the typical usage rate is 0.1-3%. While 1.5% and 2% should theoretically be sufficient with the right ions, the high levels of surfactant and glycerin might require you to use a concentration closer to the upper limit of this range, or even slightly higher, to achieve a very firm gel. You may need to experiment to find the exact concentration that gives you the desired texture.
Keep in mind that Disodium EDTA is a chelating agent, meaning it binds to metal ions. While 0.2% is a standard usage rate, in the presence of added ions for gelling, there might be some interaction. However, it's unlikely to completely inhibit gelling if sufficient potassium or sodium ions are added.
In summary, the primary reason your soap isn't gelling is likely the lack of necessary ions to activate the Kappa Carrageenan. Adding an ionic compound should resolve this. You may also need to adjust the Kappa Carrageenan concentration and consider the impact of your high surfactant and glycerin levels on the final gel firmness.