Preventing Sodium Lauroyl Glutamate Precipitation without EDTA
Asked by: bbaekhyunee0405
On: July 16, 2021
Product Type:
Cosmetics
Answered
Question
How can I prevent Sodium Lauroyl Glutamate from precipitating in a formulation without using Disodium EDTA?
Answer
Preventing Precipitation of Sodium Lauroyl Glutamate without Disodium EDTA
Precipitation of Sodium Lauroyl Glutamate in your formulation is often caused by the presence of metal ions, particularly calcium and magnesium, commonly found in tap water. These ions can react with the surfactant, forming insoluble compounds that appear as precipitate. Disodium EDTA is typically used to prevent this by chelating (binding to) these metal ions, rendering them inactive.
Since you prefer not to use Disodium EDTA, here are alternative methods and ingredients you can use:
- Use High-Purity Water: The simplest solution is to use deionized or distilled water instead of tap water. This eliminates the source of most metal ions that cause precipitation.
- Alternative Chelating Agents: Several other chelating agents can effectively bind metal ions and prevent precipitation. Consider these options:
- Sodium Citrate: This is a common and effective chelating agent that also helps control pH.
- Sodium Phytate: A natural chelating agent derived from plants. It also offers antioxidant and stabilizing properties.
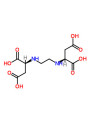
- Trisodium Ethylenediamine Disuccinate (EDDS): This is a biodegradable chelating agent known for its effectiveness.
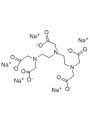
- Pentasodium Pentetate: Another effective chelating agent suitable for formulations requiring metal ion control.
- HEDP-4Na and HEDP-2Na: These are forms of Etidronic Acid salts that act as chelating agents.
- Adjust pH: Ensure your formulation's pH is within the optimal solubility range for Sodium Lauroyl Glutamate. Testing different pH levels might help improve stability.
- Incorporate Co-surfactants or Hydrotropes: Adding other compatible surfactants or hydrotropes to your formulation can sometimes enhance the solubility of Sodium Lauroyl Glutamate and prevent it from precipitating.
By implementing one or a combination of these methods, you can stabilize your Sodium Lauroyl Glutamate formulation without relying on Disodium EDTA.