Toner Formulation Stability: Addressing Sedimentation and Ball-Up with High Active Load
Question
I formulated a toner using the following composition:
- Safe B3: 8%
- Gluco Bright: 5%
- Ume Extract: 2%
- Ethoxydiglycol: 3%
- EGT-Max™: 1%
- Dipotassium Glycyrrhizate: 2%
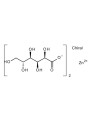
- Zinc: 1%
- Mild preservative: 1%
- Balance: Water
After mixing, the toner was initially usable. However, after standing for a period, significant sedimentation occurred at the bottom of the bottle, requiring shaking before each use. Sometimes these precipitates cause ball-up on the skin during application.
I need advice regarding the following issues:
- Should I add other solvents to solve the sedimentation problem?
- Does this precipitation result in less active ingredient being delivered to the skin?
- I have tried increasing the mixing time, but precipitation still occurs. How should this issue be fixed?
- Are there any ingredients in this formula that are difficult to dissolve? Previously, I used a formula with a higher concentration of active ingredients, but it dissolved well.
Answer
Toner Precipitation Issue
It sounds like you've created a toner with a great combination of active ingredients! The precipitation you're seeing is a common challenge in cosmetic formulation, often due to the solubility limits of the ingredients in the solvent system or interactions between different components, especially at high concentrations.
Based on the ingredients you've listed and their properties:
- Safe-B3 (Niacinamide) and GlucoBright (Acetyl Glucosamine) are generally very water-soluble and compatible, commonly used together.
- Ethoxydiglycol is a good solvent and carrier, soluble in water and glycols, which helps increase the solubility of other ingredients.
- EGT-Max (Ergothioneine) and Dipotassium Glycyrrhizate are also water-soluble. However, Dipotassium Glycyrrhizate can have a mild electrolyte nature which might affect formula stability at higher concentrations (you are using it at the upper recommended limit).
- The form of Zinc you used is crucial. If it's Zinc PCA, its solubility is highly dependent on pH, being most soluble between pH 4-6. Outside this range, it can precipitate. If it's Zinc Gluconate, while water-soluble, you are using it at a concentration (1%) that is above the typically recommended rate (0.05-0.5%), and it is also an electrolyte which can cause issues.
- The Ume Extract's specific solubility characteristics are unknown without more information, but plant extracts can sometimes contribute to precipitation.
- The high total concentration of dissolved solids (actives + Ethoxydiglycol + preservative) in the water base might be exceeding the overall solubility capacity.
Here are the answers to your questions:
1. Should we add other solvents to fix the precipitation?
Yes, adding additional co-solvents is a common way to increase the solubility of ingredients in a water-based formula like a toner. Solvents such as Propylene Glycol or Butylene Glycol are miscible with water and can help keep a higher concentration of various active ingredients dissolved. You could try adding 5-10% of one of these glycols to see if it improves the long-term stability and prevents precipitation. Increasing the percentage of Ethoxydiglycol might also help, but be mindful of the regulatory limits for leave-on products (currently around 2.6%).
2. Does this precipitation reduce the amount of active ingredient absorbed by the skin?
Yes, absolutely. When ingredients precipitate out of the solution and settle at the bottom, they are no longer evenly dispersed throughout the toner. This means that when you apply the toner, you are not getting the intended concentration of the active ingredients. The precipitated particles are also what is likely causing the "ball-up" issue on your skin, as they are solid particles rather than dissolved substances that can be absorbed.
3. Should mixing time be increased? How should this be fixed?
Increasing mixing time helps ensure that ingredients are initially dissolved and dispersed, but it will not prevent precipitation that occurs over time due to solubility limits or incompatibilities. The core issue is likely the formula's ability to keep all ingredients dissolved in the long term.
To fix this, you should consider:
- Adjusting Concentrations: Slightly reduce the percentage of ingredients that are at their upper solubility limits or above recommended usage rates, particularly the Zinc (ensure it's within the recommended range for the specific type used, e.g., 0.2-1.0% for Zinc PCA, 0.05-0.5% for Zinc Gluconate). Consider slightly reducing Dipotassium Glycyrrhizate if other measures aren't sufficient.
- Adding Co-solvents: Incorporate Propylene Glycol or Butylene Glycol into the water phase as mentioned above.
- Checking and Adjusting pH: This is critical, especially if you are using Zinc PCA. Measure the pH of your final toner. If it's outside the optimal range of 4-6 for Zinc PCA, adjust it using a mild acid (like Citric Acid solution) or base (like Sodium Hydroxide solution) to bring it into this range. A pH between 4.5 and 6.0 is often suitable for many active ingredients in toners, including Niacinamide, Acetyl Glucosamine, and Dipotassium Glycyrrhizate.
- Mixing Method: Ensure you are dissolving each ingredient thoroughly in the water phase (and co-solvents if used) before combining them. Add heat-sensitive ingredients like EGT-Max and Dipotassium Glycyrrhizate after the main water phase has cooled down (below 60°C for DPG, avoid heat for EGT-Max).
4. Are there any ingredients in the formula that are difficult to dissolve?
Yes, the most likely candidates for causing precipitation in your formula are:
- Zinc: Depending on the specific form (e.g., Zinc PCA is pH sensitive, Zinc Gluconate is used at a high concentration and is an electrolyte) and the overall formula composition, Zinc compounds are often challenging at higher percentages.
- Dipotassium Glycyrrhizate: While water-soluble, using it at 2% is the upper limit, and its electrolyte nature can contribute to instability in complex formulas.
- Ume Extract: Without knowing its specific type and processing, it might contain components that are not fully soluble or stable in the toner base.
- The combined load of all active ingredients: Even if individually soluble, putting many different ingredients at high percentages into a limited amount of solvent (water + Ethoxydiglycol) can exceed the total solubility capacity.
Focus on optimizing the solubility of the Zinc compound and potentially adding co-solvents like Propylene Glycol or Butylene Glycol while ensuring the pH is appropriate for all ingredients.