Underarm Cream Formulation Issues: Separation and Color Change
Question
I am developing an underarm cream formulation for odor control and skin whitening, but the formula is separating and changing color. Could you please provide feedback on my current formula and suggest how to prevent these issues?
My formula is as follows:
- Water: 50.8%
- Dissodium EDTA: 0.1%
- Hydroxyethyl Cellulose: 0.2%
- Polyglyceryl 10 Dipalmitate: 1.0%
- Polyglyceryl 6 Distearate: 1.0%
- Cetyl Alcohol: 5.0%
- Cetyl Dimethicone: 4.0%
- Glyceryl Stearate & PEG-100 Stearate: 1.5%
- Cyclopentasiloxane: 5.0%
- Aluminium Chlorohydrate: 15.0%
- Water: 8.0%
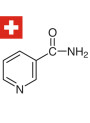
- Niacinamide: 5.0%
- Bioran Flower Acid: 1.0%
- Dipotassium Glycyrrhizate: 0.1%
- Cetetyl Morpholinium Ethosulfate: 1.0%
- Phenoxyethanol & Caprylyl Glycol & Chlorphenesin: 1.0%
- Fragrance: 0.3%
Answer
Feedback on Your Underarm Cream Formula
Thank you for sharing your underarm cream formula aimed at odor control and skin whitening. It's a comprehensive formula with several effective ingredients. Let's review the potential reasons for separation and color change and suggest some improvements.
Analysis of Your Current Formula
Your formula uses a combination of ingredients for different purposes:
- Odor Control/Antiperspirant: Aluminum Chlorohydrate is a key active for this purpose.
- Whitening: Niacinamide and potentially Bioran Flower acid are included for skin brightening.
- Emulsification & Texture: You are using multiple emulsifiers (Polyglyceryl 10 Dipalmitate, Polyglyceryl 6 Distearate, Glyceryl Stearate & PEG-100 Stearate), thickeners (Cetyl Alcohol, Hydroxyethyl Cellulose), and silicones (Cetyl Dimethicone, Cyclopentasiloxane).
- Stability & Preservation: Disodium EDTA and the preservative blend are included.
- Soothing: Dipotassium Glycyrrhizate is present to reduce irritation.
Potential Causes of Separation and Color Change
Based on your formula and the properties of the ingredients, here are some potential reasons for the instability you are experiencing:
- Complex Emulsifier System: Using a mix of W/O (Polyglyceryl 10 Dipalmitate, Polyglyceryl 6 Distearate) and O/W (Glyceryl Stearate & PEG-100 Stearate) emulsifiers in a single formula can be challenging to stabilize, especially with high water content.
- High Electrolyte Load from Aluminum Chlorohydrate: Aluminum Chlorohydrate is a highly effective antiperspirant but introduces a significant amount of electrolytes into the water phase. Many emulsifiers are sensitive to electrolytes and can cause the emulsion to break or separate. The description for Anti-Sweat™ (Aluminum Chlorohydrate) specifically mentions the need for an electrolyte-resistant emulsifier.
- pH Considerations: Aluminum Chlorohydrate is most effective and stable at a slightly acidic pH. Niacinamide is most stable between pH 4.0-7.0. If the pH of your formula is not carefully controlled within a range compatible with all key ingredients, it can lead to degradation or instability. Bioran Flower acid, if acidic, would also influence the final pH.
- Ingredient Interactions: The combination of various ingredients, including different emulsifiers, high electrolyte content, and potentially acidic components, can lead to complex interactions that destabilize the emulsion or cause color changes over time.
- Dipotassium Glycyrrhizate: While beneficial, its description notes it has a mild electrolyte nature and can potentially make a formula slightly more liquid, which might contribute to separation if the emulsion is already borderline stable.
Suggestions for Improvement
To improve the stability and prevent separation and color change, consider the following adjustments:
- Simplify the Emulsifier System: Focus on using one or a blend of O/W emulsifiers that are known to be robust and tolerant to high electrolyte concentrations and slightly acidic pH levels. Glyceryl Stearate and PEG-100 Stearate is a good candidate as it is noted as electrolyte resistant, but ensure the overall emulsifier system is balanced for your specific oil and water phases and the high active load.
- Optimize Manufacturing Process: Ensure your heating, mixing (especially the homogenization step), and cooling processes are appropriate for forming a stable emulsion with the chosen ingredients.
- Carefully Control pH: Adjust the final pH of your formula to a range that ensures the stability and efficacy of both Aluminum Chlorohydrate and Niacinamide, likely in the slightly acidic range (e.g., pH 4.0-5.0), provided your emulsifier system can handle this pH.
- Check Ingredient Compatibility: If issues persist, review the compatibility of each ingredient, particularly the cationic Cetetyl Morpholinium Ethosulfate, with the rest of the formula components under the target pH and electrolyte conditions.
By addressing the emulsification system, managing the electrolyte load from Aluminum Chlorohydrate, and carefully controlling the pH, you should be able to create a more stable underarm cream that does not separate or change color while providing both odor control and whitening benefits. Continue testing small batches with adjustments to find the optimal balance for your formula.