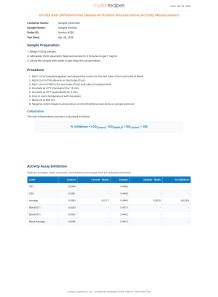
Antitrypsin Activity Measurement
This antitrypsin activity assay is important because it measures the ability of a sample (e.g., plant extract) to inhibit the proteolytic enzyme trypsin, which is crucial in many biological and industrial contexts (e.g., therapeutic enzyme modulation, functional food ingredient evaluation). The method uses BApNA, a chromogenic substrate that releases a yellow-colored product (p-nitroaniline) upon hydrolysis by trypsin. By comparing absorbances of control vs. sample, one can calculate the percentage inhibition of trypsin activity. Tris-HCl buffer is used instead of phosphate buffer to avoid unwanted buffer-catalyzed BApNA hydrolysis, ensuring reliable and accurate measurement.
Sample Count:
Accepts exactly 1 sample.
Anti-Inflammation
Anti-Inflammation analysis
UV-VIS Anti-Inflammation (Based-on Protein Denaturation) Activity Measurement
Egg albumin is employed as a model protein in the experiment, and denaturation is brought about by exposing it to extremes of heat, pH, or other denaturing agents. Egg albumin’s original conformation is disrupted during denaturation, changing its physical characteristics, and causing it to lose its functional activity. The egg albumin denaturation assay measures a drug or compound’s capacity to prevent or lessen egg albumin denaturation to evaluate its anti-inflammatory effects. The egg albumin denaturation assay is based on the idea that substances with anti-inflammatory qualities may be able to stabilize protein structures and prevent denaturation, which is frequently linked to inflammation and tissue damage. As a result, agents or chemicals that significantly decrease the denaturation o
Protein denaturation inhibition
The protein denaturation inhibition assay is essential for evaluating the anti-inflammatory properties of plant extracts and compounds. By assessing the ability of a sample to prevent or reduce the denaturation of egg albumin, researchers can identify potential anti-inflammatory agents that stabilize protein structures, thereby mitigating inflammation and tissue damage. This assay is valuable in pharmaceutical development, natural product research, and the formulation of anti-inflammatory therapies. The assay employs egg albumin as a model protein whose denaturation is induced by heat. Anti-inflammatory agents are hypothesized to stabilize the protein structure, preventing denaturation. By measuring the extent of albumin denaturation through absorbance at 660 nm, the method provides a reli
Cart
No products
Subtotal:
0.00
Total
0.00
THB
Loading Researcher Network details...