Determination of Carbamide Peroxide Content by Iodometric Titration Method
Iodometric titration method determines the carbamide peroxide (CP) content in a sample (crystals or gel) by generating iodine in an acidic medium containing potassium iodide and a catalyst (ammonium molybdate). The liberated iodine is titrated with sodium thiosulfate, and the CP content is calculated from the volume of titrant used. Carbamide peroxide is widely used in dental, cosmetic, and pharmaceutical formulations as a bleaching and antiseptic agent. Accurate quantification ensures product efficacy, safety, and regulatory compliance. Under acidic conditions, carbamide peroxide oxidizes iodide to iodine. The amount of iodine generated is stoichiometrically proportional to the CP in the sample. Titration of iodine with standardized sodium thiosulfate provides a reliable measure of CP con
Services
Analytical Services for cosmetics, personal care products
Determination of total Antioxidant Activity using DPPH Assay
Antioxidant activity was measured with the DPPH (2,2-diphenyl-1-picrylhydrazyl) assay by UV-Vis spectrophotometer which is a common method to evaluate the ability of compounds to scavenge free radicals. This assay was widely applied to assess the antioxidant properties of natural products, extracts, and compounds.
Sample Count:
Accepts exactly 1 sample.
Folin-Ciocalteu Assay for Total Phenolic Content (UV-Vis)
Spectrophotometric method measuring total phenolics in plant extracts via reaction with Folin-Ciocalteu reagent, absorbance at 765 nm, expressed as gallic acid equivalents.
Sample Requirement:
plant extract (lyophilized or dry) · 0.1 · grams
Sample Count:
Accepts exactly 1 sample.
Franz Cell Analysis with Porcine Skin Permeation Report
The porcine skin permeation test using a Franz diffusion cell is a method commonly used in pharmaceutical and cosmetic research to evaluate the permeation of substances through porcine skin. It provides valuable information on the transdermal delivery potential of pharmaceutical and cosmetic products and aids in the development of formulations with enhanced skin permeability.
Sample Count:
Accepts between 1 and 6 samples.
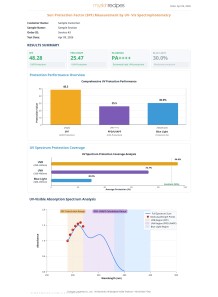
Sun Protection Factor (SPF) Measurement by UV- Vis Spectrophotometry
Experiment to test the SPF and PA value for active sample to determine the sunburn protection rate on human skin.
Sample Requirement:
Take a sample volume of 30 grams. The sample must be homogeneous and not separated.
Sample Count:
Accepts exactly 1 sample.
Cart
No products
Subtotal:
0.00
Total
0.00
THB
Loading Researcher Network details...