Determination of Carbamide Peroxide Content by Iodometric Titration Method
- Product Code: 31997
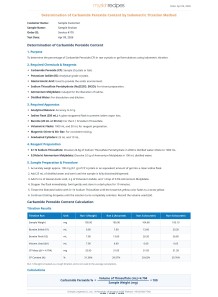
Iodometric titration method determines the carbamide peroxide (CP) content in a sample (crystals or gel) by generating iodine in an acidic medium containing potassium iodide and a catalyst (ammonium molybdate). The liberated iodine is titrated with sodium thiosulfate, and the CP content is calculated from the volume of titrant used. Carbamide peroxide is widely used in dental, cosmetic, and pharmaceutical formulations as a bleaching and antiseptic agent. Accurate quantification ensures product efficacy, safety, and regulatory compliance. Under acidic conditions, carbamide peroxide oxidizes iodide to iodine. The amount of iodine generated is stoichiometrically proportional to the CP in the sample. Titration of iodine with standardized sodium thiosulfate provides a reliable measure of CP content based on well-established redox chemistry.
description Sample Requirement
Sample Mass= 0.1g (per titration)
Replication: Quadruplicate
Total Sample Required: 0.4g
insert_drive_file Report of Previously Tested Sample
The report will be available soon. If you need it sooner, please contact customer service.
timeline Service Steps
| Step | Procedure | Expected Result |
|---|---|---|
|
1
|
Determination of... |
check_circle_outline
- |
|
2
|
2. Required... |
check_circle_outline
- |
|
3
|
3. Required... |
check_circle_outline
- |
|
4
|
4. Reagent... |
check_circle_outline
|
|
5
|
5. Sample... |
check_circle_outline
|
|
6
|
2. Add 25... |
check_circle_outline
|
|
7
|
3. Add... |
check_circle_outline
|
|
8
|
4. Stopper the... |
check_circle_outline
|
|
9
|
5. Titrate the... |
check_circle_outline
|
Cart
No products
Subtotal:
0.00
Total
0.00
THB
Loading Researcher Network details...