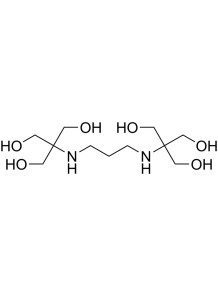
BIS-TRIS Propane (BTP)
also known as 1,3-Bis(tris(hydroxymethyl)methylamino)propane, is a chemical compound that is commonly used as a buffer in biochemical and molecular biology applications
BIS-TRIS Propane, also known as 1,3-Bis(tris(hydroxymethyl)methylamino)propane, is a chemical compound that is commonly used as a buffer in biochemical and molecular biology applications. Here are some key details about BIS-TRIS Propane (BTP):
1. Chemical Structure and Properties:
- BTP is a zwitterionic buffer compound, containing both positively and negatively charged groups within the same molecule.
- It is a white, crystalline powder that is highly soluble in water and other polar solvents.
- BTP has a pKa value around 6.8, making it useful for maintaining a slightly acidic to neutral pH range.
2. Applications:
- Protein Purification: BTP is often used as a buffer component in protein purification procedures, such as ion-exchange chromatography and affinity chromatography.
- Enzyme Assays: BTP buffers can help maintain the optimal pH environment for various enzymatic reactions and activity assays.
- Nucleic Acid Manipulation: BTP can be used as a buffer in applications involving DNA and RNA, such as gel electrophoresis and enzymatic reactions.
- Cell Culture: BTP may be included in cell culture media to help maintain the desired pH range for cell growth and metabolism.
3. Advantages:
- pH Buffering Range: BTP is effective in maintaining a pH range of approximately 6.0-8.0, making it suitable for applications that require a slightly acidic to neutral environment.
- Compatibility: BTP is generally considered a biocompatible buffer and is widely used in biological and biochemical applications.
- Solubility: BTP has good solubility in water and other polar solvents, allowing for easy preparation of buffer solutions.
4. Considerations:
- Temperature Dependence: The pH of BTP buffer solutions can be affected by temperature, so the pH should be carefully monitored and adjusted as needed.
- Potential Interactions: Like any buffer, BTP may interact with certain compounds or materials, so its compatibility should be evaluated for specific applications.
Overall, BIS-TRIS Propane (BTP) is a versatile and commonly used buffer in various biochemical and molecular biology applications, particularly in the areas of protein purification, enzyme assays, and nucleic acid manipulation.
- Cool (15-25C)
- 24 Months from manufacturing or testing date.
- Add into water phase. Heat tolerant.
- Avoid heat above 80C
- 3.00 - 7.00
- Soluble in Water, Soluble in glycols, Soluble in Hydroalcoholic Solutions
- -
| Test Name | Specification |
|---|---|
| Appearance | White crystalline powder |
| Assay (%) | 99-101 |
| Melting Point (C) | 162-165 |
| Solubility (0.1M aq.) | Clear, colorless solution |
| Loss on drying (%) | 1 Max |
| pH (1% aq.) | 10.4-11.0 |
| Ultraviolet Absorbance (0.1M aq., 280nm) | 0.15 Max |
| Ultraviolet Absorbance (0.1M aq., 260nm) | 0.05 Max |
Cart
No products