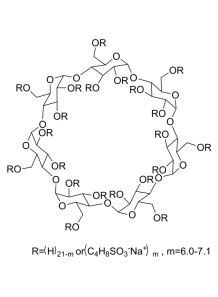
Sulfobutylether-β-cyclodextrin (SBE-β-CD)
modified form of the cyclic oligosaccharide beta-cyclodextrin. It is widely used for several applications due to its unique properties
Sulfobutylether-β-cyclodextrin (SBE-β-CD) is a modified form of the cyclic oligosaccharide beta-cyclodextrin. It is widely used for several applications due to its unique properties. Here are some key points about the usage of SBE-β-CD:
Solubilization of poorly soluble drugs: SBE-β-CD can significantly improve the aqueous solubility of poorly water-soluble drugs by forming inclusion complexes with the drug molecules. This property is extensively exploited in the pharmaceutical industry to formulate drugs with low solubility.
Parenteral drug delivery: SBE-β-CD is relatively non-toxic and can be administered intravenously or intramuscularly. It is used as a solubilizing agent for parenteral formulations of drugs with low solubility.
Stabilization of drugs: SBE-β-CD can protect drugs from degradation by encapsulating them within its hydrophobic cavity, shielding them from factors like light, oxidation, and hydrolysis.
Masking bitter taste: SBE-β-CD can form inclusion complexes with bitter-tasting drugs, masking their unpleasant taste, which is beneficial for oral formulations.
Enzyme immobilization: SBE-β-CD can be used as a support material for immobilizing enzymes, improving their stability and allowing their reuse in various industrial processes.
Analytical chemistry: SBE-β-CD is used in analytical techniques like capillary electrophoresis and chromatography for separating and analyzing various compounds.
Environmental remediation: SBE-β-CD can form inclusion complexes with environmental pollutants like polycyclic aromatic hydrocarbons and organochlorine pesticides, aiding in their removal from contaminated sites.
- Room (25-40C)
- 24 Months from manufacturing or testing date.
- 0.00% - 0.00%
- 0.00%
- Add into water phase. Heat tolerant.
- Heat Tolerant
- 0.00 - 0.00
- Soluble in Water, Soluble in Ethanol, Dispersible in water, Soluble in DMSO (Dimethyl Sulfoxide)
- Solubilizing Agent, Pharmaceutical Excipient
- -
| Test Name | Specification |
|---|---|
| Appearance | White to off-white amorphous powder |
| Solubility | Freely soluble in water, sparingly soluble in ethanol, practically insoluble in ethanol, in n-hexane, in 1-butanol, in acetonitr |
| Identification: IR | Same absorption bands as USP betadex sulfobutyl ether sodium RS |
| Identification: HPLC | The retention time of the major peak of sample solution corresponds to the standard solution |
| Identification: Average degree of substitution | Conform to standard |
| Identification: Sodium | Identify test as positive for sodium |
| Assay (on the anhydrous basis) | 95.0-105.0% |
| Betadex | 0.1% Max |
| 1,4-butane sultone | 0.5ppm Max |
| Sodium chloride | 0.2% Max |
| 4-hydroxybutane-1-sulfonic acid | 0.09% Max |
| Bis(4-sulfobutyl) ether disodium | 0.05% Max |
| Bacterial endothoxins | 0.01EU/mg Max |
| The total aerobic microbial count | 100CFU/g Max |
| The total combined molds and yeasts count | 50CFU/g Max |
| E.Coli | Absent |
| Average degree of substitution | 6.2-6.9 |
| Peaks 1 | 0.0-0.3 |
| Peaks 2 | 0.0-0.9 |
| Peaks 3 | 0.5-5.0 |
| Peaks 4 | 2.0-10.0 |
| Peaks 5 | 10.0-20.0 |
| Peaks 6 | 15.0-25.0 |
| Peaks 7 | 20.0-30.0 |
| Peaks 8 | 10.0-25.0 |
| Peaks 9 | 2.0-12.0 |
| Peaks 10 | 0.0-4.0 |
| pH | 4.0-6.8 |
| Water determination | 10% Max |
| Clarity of solution | 30% (w/v) solution is clear and essentially free from particles of foreign matter |
Cart
No products