Methanol (99.9%)
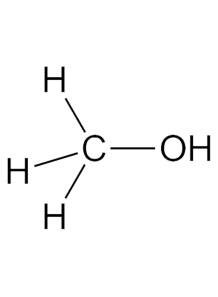
often called wood alcohol, is the simplest alcohol with the chemical formula CH₃OH. It is a colorless, volatile, and flammable liquid at room temperature, with a characteristic faintly sweet odor
Methanol, often called wood alcohol, is the simplest alcohol with the chemical formula CH₃OH. It is a colorless, volatile, and flammable liquid at room temperature, with a characteristic faintly sweet odor. Methanol is produced both naturally and synthetically, and it has numerous industrial and commercial applications.
Key Properties:
- Chemical Formula: CH₃OH
- Molecular Weight: 32.04 g/mol
- Physical State: Liquid at room temperature
- Boiling Point: Approximately 64.7 °C (148.5 °F)
- Solubility: Completely miscible with water, ethanol, ether, and many other organic solvents
Production:
Historically, methanol was produced by the destructive distillation of wood, giving rise to the name “wood alcohol.” Today, it is predominantly synthesized from syn-gas (a mixture of carbon monoxide and hydrogen) using a copper-based catalyst. Another emerging method involves converting methane (the primary component of natural gas) into methanol, representing a key component in natural gas-to-liquids (GTL) technologies.
Uses and Applications:
- Chemical Feedstock: Methanol is a crucial starting material for manufacturing a wide range of chemicals, including formaldehyde, acetic acid, methyl tertiary-butyl ether (MTBE), and various plastics, resins, and adhesives.
- Fuel and Energy Applications: Methanol can be blended with gasoline to create high-octane fuel blends, and it’s used as a fuel in specialized racing engines. In addition, there’s growing interest in using methanol as a hydrogen carrier for fuel cells and for powering direct methanol fuel cells (DMFCs).
- Solvent and Extraction Agent: Thanks to its polarity and ability to dissolve a broad range of substances, methanol is commonly used as a solvent in laboratories and industrial processes.
- Denaturant for Ethanol: Methanol is frequently added to ethanol to make it undrinkable, a process known as denaturing, thus avoiding beverage alcohol taxes.
- Cool (15-25C)
- 24 Months from manufacturing or testing date.
- Add at last step. Avoid temperature above 40C.
- Avoid heat above 80C
- 0.00 - 0.00
- Soluble in Water, Soluble in glycols, Soluble in Ethoxydiglycol, Soluble in Ethanol, Soluble in DMSO (Dimethyl Sulfoxide)
- Cleanser
- -
| Test Name | Specification |
|---|---|
| ASSAY | 97–100 |
| CA | 0–200 |
| CD | 0–50 |
| CO | 0–50 |
| Cu | 0–50 |
| K | 0–100 |
| IRON(FE) | 0–50 |
| Melting point | 60–70 |
| NI | 0–50 |
| PB | 0–50 |
| ZN | 0–50 |
| X-RAY DIFFRACTION | Conforms to Structure |
| Appearance | WHITE POWDER OR CRYSTALS |
Cart
No products