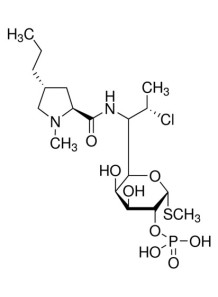
Clindamycin Phosphate
Used in acne medications to control P. Acnes bacteria, which is the cause of acne inflammation.
Grade USP 43, order size 25kg
This product can be sold only to hospitals, clinics, pharmaceutical factories, pharmacists, and doctors for research purposes only. The buyer must submit the following documents: Individuals: Medical or pharmacy professional certification documents. Legal entities: Drug manufacturing facility certification documents or hospital (clinic/hospital) certification documents.
Clindamycin Phosphate is used in acne medications to control P. Acnes, the bacteria that causes acne.
Use: Used in medicine only. Not allowed to be used in cosmetics.
Product appearance: white/off-white powder
Storage: For long-term storage, keep in the refrigerator, avoid sunlight and heat, seal the lid tightly, shelf life is at least 2 years.
Chemical Name : Clindamycin Phosphate
- Cool (15-25C)
- 24 Months from manufacturing or testing date.
- Please follow instruction under product info carefully.
- Heat Tolerant
- 0.00 - 0.00
- Soluble in Water, Soluble in Ethanol, Soluble in DMSO (Dimethyl Sulfoxide)
- -
| Test Name | Specification |
|---|---|
| Appearance | White to off-white, hygroscopic, crystalline powder |
| Identification (IR) | The IR absorption spectrum is consistent with the standard spectrum. |
| Identification (HPLC) | The retention time of the main peak in the HPLC sample solution is the same as that in the standard solution |
| pH | 3.5-4.5 |
| Water Content | 6.0% Max |
| Crystallinity | Meet the requirement |
| Assay | Min 758 |
| Bacterial Endotoxins | Max 0.58EU/mg |
| Lincomycin phosphate | 1.0% Max |
| Lincomycin | 0.5% Max |
| Clindamycin B phosphate | 1.5% Max |
| 7-Epiclindamycin phosphate | 0.8% Max |
| Clindamycin 3-phosphate | 0.3% Max |
| Clindamycin | 0.5% Max |
| Other impurity | 1.0% Max |
| Total impurities | 4.0% Max |
Cart
No products