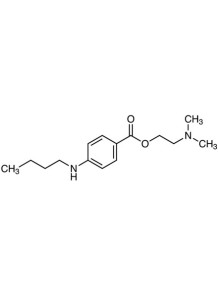
Tetracaine (Base)
Tetracaine (Base) Used in ointments to numb the skin.
This product Can only be sold to hospitals, clinics, pharmaceutical factories, pharmacists, and doctors for use in research. The buyer must submit the following documents: Natural person: Medical or pharmacist professional certification document. Legal entity: Document to receive information about the drug production location. or hospital certification document (Clinic/Hospital)
Tetracaine (Base) Used in ointments to numb the skin.
Usage: Use only in medicine Not allowed for use in cosmetics.
Product characteristics: white flake powder
Storage: If you want to keep it for the long term Store in the refrigerator. Do not expose to sunlight or heat. Seal the lid tightly. Shelf life is at least 2 years.
- Cool (15-25C)
- 24 Months from manufacturing or testing date.
- Please follow instruction under product info carefully.
- Heat Tolerant
- 3.50 - 7.00
- Soluble in Ethanol, Soluble in DMSO (Dimethyl Sulfoxide)
- Topical Anesthetic, Pharmaceutical Intermediate
- -
| Test Name | Specification |
|---|---|
| Appearance | White or light yellow waxy solid |
| Solubility | Very slightly soluble in water; soluble in ethanol, in ether, in benzene and in chloroform |
| Melting point | Form 41°C to 46°C |
| IR | Complies |
| HPLC | The retention time of the major peak of the sample solution corresponds to that of the standard solution dissolved in the Assay |
| Aminobenzoic acid | Not more than 0.10% |
| Impurity B | Not more than 0.10% |
| Impurity C | Not more than 0.10% |
| Individual unspecified impurity | Not more than 0.10% |
| Total impurities | Not more than 0.50% |
| Ethanol | Not more than 0.50% |
| Acetone | Not more than 0.50% |
| Toluene | Not more than 0.089% |
| Isopropyl ether | Not more than 0.50% |
| Loss on drying | Not more than 0.50% |
| Residue on ignition | Not more than 0.10% |
| Assay | NLT 98.0% and NMT 102.0% of C15H24N2O2, calculated on the dried basis |
Cart
No products